Summary
DOI: 10.1373/clinchem.2010.160994
A 62-year-old woman presenting with dyspnea on exertion was admitted to our hospital. Her medical history included type 2 diabetes and κ light chain multiple myeloma (MM) diagnosed 3 years ago.
Student Discussion
Student Discussion Document (pdf)
Jean-Baptiste Oudart,1 François-Xavier Maquart,1,2 Oualid Semouma,1 Magali Lauer,1
Patricia Arthuis-Demoulin,1 and Laurent Ramont1,2*
1CHU de Reims, Laboratoire Central de Biochimie, Reims, France; 2Université de Reims Champagne-Ardenne, CNRS UMR 6237, Reims, France.
*Address correspondence to this author at: Laboratoire Central de Biochimie, CHU de Reims, Rue Serge Kochman, 51092 Reims Cedex, France. Fax: +0033-326788539; E-mail: [email protected].
Case Description
A 62-year-old woman presenting with dyspnea on exertion was admitted to our hospital. Her medical history included type 2 diabetes and light chain multiple myeloma (MM) diagnosed 3 years ago.
The results of a basic biochemical examination performed 2 weeks earlier were normal. Serum protein electrophoresis revealed hypoproteinemia (65 g/L; reference interval, 68–73 g/L), associated with decreased γ globulins (4 g/L; reference interval, 9–15 g/L) but without a detectable paraprotein band. An assay for serum free light chain showed decreased κ light chains (0.5 mg/L; reference interval, 3.3–19.4 mg/L) and λ light chains (<0.3 mg/L; reference interval, 5.7–26.3 mg/L); the κ/λ ratio could not be accurately determined because of the low concentration of λ light chains. An examination of a bone marrow aspirate taken 3 months earlier showed dystrophic plasma cells accounting for 50% of the nucleated cells.
On admission, laboratory tests revealed normal values for hemoglobin (142 g/L; reference interval, 130–180 g/L), white blood cells (6.7 X 109/L; reference interval, 4–10 X 109/L), platelets (211 X 109/L; reference interval, 150–400 X 109/L), and creatinine [38 μmol/L (0.4 mg/dL); reference interval, 45–90 μmol/L (0.5–1.0 mg/dL)], but the tests also revealed mild hypocalcemia [2.14 mmol/L (8.6 mg/dL); reference interval, 2.20–2.60 mmol/L (8.8–10.4 mg/dL)].
A chest radiograph and spiral thoracic computed tomography showed a large right-sided pleural effusion.
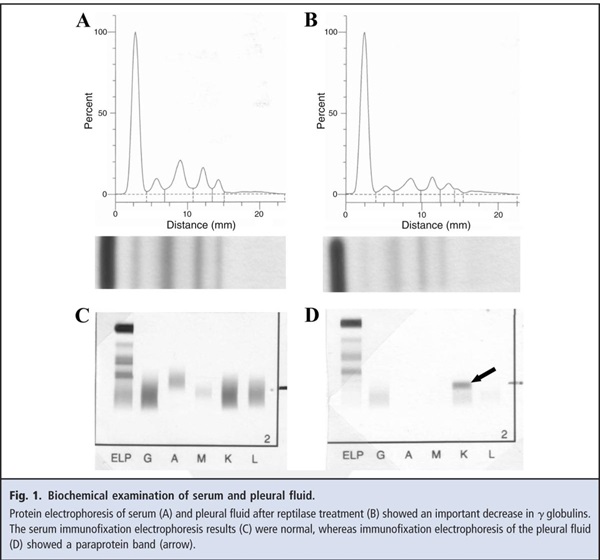
A sample of the pleural fluid had a total protein concentration of 38 g/L and a white blood cell count of 20.5 X 109/L, with 100% lymphoid cells. The results of bacterial and mycobacterial cultures were negative. Protein electrophoresis evaluations of serum, urine, and the pleural effusion were performed in our laboratory with the Hydrasys® electrophoresis system (Sebia). The serum protein electrophoresis results confirmed the hypoproteinemia (total serum protein, 51 g/L), which was associated with a markedly decreased γ-globulin concentration (2 g/L). There was no detectable paraprotein band (Fig. 1A). The urine electrophoresis results showed proteinuria (713 mg/24 h; reference interval, <150 mg/24 h) associated with 2 bands that migrated in SDS-PAGE at the positions of free light chains (data not shown). Standard electrophoresis analysis of the unconcentrated pleural effusion showed a discrete band migrating at a position between the β and γ globulins. This band corresponded to the presence of residual fibrinogen (1), because it disappeared after reptilase treatment (Fig. 1B).
Reference
1. Qiu LL, Levinson SS, Keeling KL, Elin RJ. Convenient and effective method for removing fibrinogen from serum specimens before protein electrophoresis.
Clin Chem 2003;49:868–72.
Questions to Consider
- What is the differential diagnosis of a myelomatous pleural effusion in MM?
- Should laboratory testing of pleural fluid in patients with myeloma include immunofixation electrophoresis?
- Must measurement of pleural fluid for free light chains be considered in this patient?
Final Publication and Comments
The final published version with discussion and comments from the experts appears
in the April 2012 issue of Clinical Chemistry, approximately 3-4 weeks after the Student Discussion is posted.
Educational Centers
If you are associated with an educational center and would like to receive the cases and
questions 3-4 weeks in advance of publication, please email [email protected].
AACC is pleased to allow free reproduction and distribution of this Clinical Case
Study for personal or classroom discussion use. When photocopying, please make sure
the DOI and copyright notice appear on each copy.
DOI: 10.1373/clinchem.2010.160994
Copyright © 2012 American Association for Clinical Chemistry