Protein electrophoresis has changed significantly since its initial development in the 1930s by Tiselius to the current gel and capillary-based methods. Along with the technology improvements, a narrowing of its clinical utility has also occurred. Its main clinical utility now is identification and monitoring of monoclonal gammopathy. Surprisingly, unlike many clinical biochemistry tests, little work has been done on standardizing protein electrophoresis reporting.
I began my career 15 years ago in a laboratory with a wealth of knowledge and experience, the next most junior Biochemist had 25 years of experience. When integrating into the lab, in particular reporting protein electrophoresis, I was instantly standardized to the well honed reporting that had been developed over many years with cooperation from clinical hematologists. All was well and I never really thought there was need for standardizing PE reporting…that is until our laboratory had a large turnover of Biochemists. We hired 3 bright young Biochemists all with varied experience in protein electrophoresis. Before long, I realized each of us had a distinct way of interpreting and reporting PE and there was a great need to standardize within our institution. This was the beginning of our Canadian standardization effort (1).
We had many laboratory discussions on how to internally (re)standardize our reporting and heavily engaged our clinical hematologists, since they (and ultimately the patient) are the ones who we are standardizing for. Together (Biochemists and Hematologists), we arrived at a consensus on how PE results should be reported. It went so smooth that we decided to engage the rest of Canada and produce a Canadian consensus on PE reporting. We generated a list of PE-related standardization topics and a suggested starting point for discussion on each topic.
One aspect that was identified early on was the need for standardization of nomenclature. Over the years there have been innumerable names for a monoclonal protein, e.g. M-spike, M-band, M-protein, monoclonal component, extragradient, and the list goes on. Tate et al (2) in their Recommendations for standardized reporting of protein electrophoresis in Australia and New Zealand proposed a limited nomenclature set, as did we. Our inaugural meeting, which included pan-Canada representation of Biochemists from academic health science centres, community hospital and private laboratories, began with a discussion of nomenclature. It seemed like a reasonable place to start, since moving to more complex issues like synoptic reporting, required we agree upon nomenclature. The initial list was posted, and the “discussion” began! Two hours later I think we had actually somehow regressed and had not agreed upon any nomenclature. Although we unable to achieve consensus on specific nomenclature, it was clear that all involved did agreed on the need to standardize nomenclature, and indeed the need to standardize all aspects of PE reporting. Many were able to come to agreement, however a few individuals refused to give up their beloved terminology. A 2017 survey of Clinical and Medical Biochemists in Canada showed there was a clear majority that agreed on the need for common nomenclature and reporting.
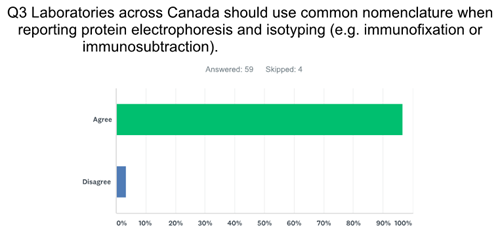
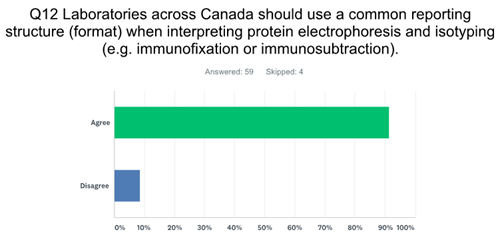
There is no direct evidence in the literature that a lack of standardization causes harm to patients, but I can tell you that I have had numerous conversations with clinical hematologists who do not understand how to interpret certain PE reports, in many cases due to the use of non-standardized terminology. I suspect many of you reading this have had similar discussions! This delay in accurate interpretation or possibly misinterpretation clearly may have negative clinical consequences.
The genetics and protein nomenclature communities, which are far more complex than PE, are advanced in the standardization of nomenclature, so why cant we standardize? Why are we so attached to our historical names for a monoclonal protein? I’m sure there were “winners” and “losers” in the genetics and protein name-game, and there will be with PE as well. But really, don’t we all win when we speak the same language?
Give up your beloved nomenclature and long held traditions and join us in our journey to improved laboratory medicine by standardizing protein electrophoresis!
1. Booth, R. A., McCudden, C. R., Balion, C. M., Blasutig, I. M., Bouhtiauy, I., Rodríguez-Capote, K., et al. (2018). Candidate recommendations for protein electrophoresis reporting from the Canadian Society of Clinical Chemists Monoclonal Gammopathy Working Group. Clinical Biochemistry, 51, 10–20. h
2. Tate, J., Caldwell, G., Daly, J., Gillis, D., Jenkins, M., Jovanovich, S., et al. (2012). Recommendations for standardized reporting of protein electrophoresis in Australia and New Zealand. Annals of Clinical Biochemistry, 49(3), 242–256.