This Scientific Short has been updated. To access the latest article click here.
It is well recognized that a majority of lab testing errors occur in the preanalytical phase prior to the blood sample being placed onto the laboratory analyzer (1). The frequency at which such errors have been reported is summarized in the following figure, adapted from Cararro et. al. (2).
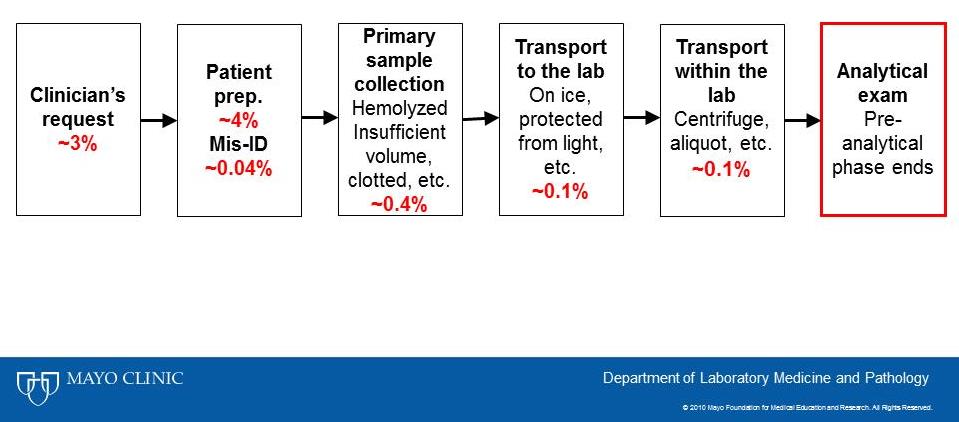
The challenges that face the laboratory include identifying potential errors that occur outside of their four walls that are unseen and may result from practices they have little control over. It is well recognized that fewer errors occur when dedicated phlebotomists collect blood compared to multi-tasking clinical staff (3).
Laboratories have gotten creative by using various mechanisms to detect potential pre-analytical problems for which they can investigate and request a redraw when needed. These solutions include providing decision support at electronic order entry to guide test utilization, automated analytical systems to identify hemolyzed and clotted specimens, and middleware rules to look for irregular patterns of patient results (i.e. including, but not limited to, delta checks) that can signify a problem. Some of these systems detect errors reliably, while the efficiency of others are not well documented and may depend on the specifics of the practice and patient population served.
What actions can be taken to prevent pre-analytical errors? There are a few measures to consider including development of clear SOPs, a rigorous training program, and automating/standardizing the process as much as possible so that doing the right thing reproducibly is easy and becomes second nature. In addition, developing quality indicators that can be used as an objective means to monitor consistency across settings and over time will help signal a problem. Ideally, all steps of the pre-analytical phase would be monitored including appropriateness of test requests, completeness of order requisition and information needed to perform testing, mis-IDs, specimens lost or damaged in transport, specimens hemolyzed, clotted, having insufficient volume, and improperly labeled or stored specimens 1. It’s important to set appropriate limits and take action when they aren’t being met.
An event reporting system that identifies areas for improvement is an important addition to the lab’s arsenal for error detection. Most labs are required to have one and most institutions utilize them as well. Complaints originate from both inside and outside of the clinical pathology department (both are important to investigate) and can identify common themes, allow determination of root cause, and corrective measures can be taken whenever possible. For all cases, it is important to close the loop with whomever submits the complaint. Acknowledging a problem and communicating the follow-up goes a long way toward improving customer satisfaction, even if the problem can’t be resolved or improved in a timely manner. It is important to beware of the difference between perceived errors and perceived causes of errors compared to data from root cause analysis because perceptions can often be very different from reality. It is well worth the effort to collect data, even in a manual fashion if needed, to establish that a problem indeed exists and identify its cause; you may discover that the cause is what you least expect.
In an upcoming AACC Management Series webinar, “Data Driven Solutions for Common Pre-Analytical Problems”, several case studies for process improvement projects will be explored that highlight the power of using data to make decisions and improve quality.
REFERENCES
- Plebani M. Quality indicators to detect pre-analytical errors in laboratory testing. Clin Biochem Rev. 2012;33:85-8.
- Carraro P, Zago T, Plebani M. Exploring the initial steps of the testing process: Frequency and nature of pre-preanalytic errors. Clin Chem. 2012;58:638-42.
- Sheppard C, Franks N, Nolte F, Fantz C. Improving quality of patient care in an emergency department: A laboratory perspective. Am J Clin Pathol. 2008;130:573-7.