The following post was written to update a previous Short. Please see the past Short on this topic here.
Diagnosing diabetes mellitus in 2021
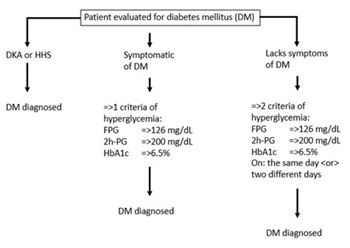
Diabetes mellitus (DM) is a family of disorders characterized by chronic hyperglycemia and the subsequent development of long-term macrovascular, microvascular and/or neuropathic complications. The 2021 American Diabetes Association (ADA) clinical practice recommendations for the diagnosis of DM are consistent with the 2020 guidelines [1]. There are 3 common scenarios where DM is diagnosed: [1] Hyperglycemic crisis; [2] DM is diagnosed in a symptomatic patient, and [3] DM is diagnosed in an asymptomatic patient (Figure 1).
Hyperglycemia crises in the diagnosis of diabetes mellitus
DM is diagnosed if a person presents with a hyperglycemic crisis: either diabetic ketoacidosis (DKA) or hyperglycemic hyperosmolar state (HHS). The table below comparing and contrasting DKA and HHS is adapted from the ADA’s 2004 guidelines [2].
|
DKA |
HHS |
| Plasma glucose |
>250 mg/dL |
>600 mg/dL |
| Arterial pH |
=<7.3 |
>7.3 |
| Serum bicarbonate |
=<18 meq/L |
>15 meq/L |
| Urine ketones |
Positive |
Small |
| Serum ketones |
Positive |
Small |
| Effective serum osmolality |
Variable |
>320 mosm/L |
| Anion gap |
=>10 meq/L |
Variable |
| Mental status |
Alert – coma |
Stupor - coma |
Note: Effective serum osmolality pertains only to sodium (Na+) and glucose [(2 x [sodium (meq/L))] + [(glucose (mg/dL)]/18)] because urea passes freely across cell membranes and does not affect the distribution of water between the intracellular and the extracellular compartments. Anion gap is the difference between the major extracellular cation sodium (Na+) and the two major extracellular anions chloride (Cl-) and bicarbonate (HCO3-). Note also that this table does not include modern measurements of serum ketones, specifically, the measurement of beta-hydroxybutyrate suggesting that these descriptions should be updated.
While there is overlap between many of the features of these 2 conditions, pH would appear to best discriminate DKA and HHS. Hyperosmolality by itself does not differentiate DKA and HHS. Not listed in the table is that DKA is most common in type 1 diabetes (T1D) and HHS is most common (although infrequent) in type 2 diabetes (T2D). About one third of children with new-onset T1D present in DKA. However, it is uncommon that adults with new-onset T2D present with HHS. T1D patients tend to be younger and lean whereas T2D patients are usually age 40 or older, and overweight or obese. Despite these generalities, about 25% of cases of T1D present after age 18 years and T2D is becoming increasingly common in children and adolescents [3].
Diagnosing diabetes in patients symptomatic of diabetes in the absence of a diabetic crisis
In persons who present with classic symptoms consistent with DM (e.g., polyuria, polydipsia and/or weight loss), hyperglycemia or an elevated hemoglobin A1c (HbA1c) only need be recognized on a single occasion to establish the diagnosis of DM. The table below summarizes the criteria for hyperglycemia.
|
Reference interval |
Hyperglycemia/elevated |
| Fasting plasma glucose (FPG) |
70 – 99 mg/dL
(3.9 – 5.5 mmol/L)
|
=>126 mg/dL
(=>7.0 mmol/L)
|
| 2-hour plasma glucose |
<140 mg/dL |
=>200 mg/dL |
| during an OGTT (2h-PG) |
(<7.8 mmol/L) |
(=>11.1 mmol/L) |
| HbA1c |
<5.7%
(<39 mmol/mol)
|
=>6.5%
(=>48 mmol/mol)
|
OGTT = oral glucose tolerance test.
Note that for each parameter [(FPG: 100-125 mg/dL (5.6 – 6.9 mmol/L); 2h-PG: 140-199 mg/dL (7.8 – 11.05 mmol/L) and; HbA1c: 5.7-6.4% (39 – 46 mmol/mol)], there are gray zones consistent with prediabetes when DM is not otherwise diagnosed [4]. Prediabetes itself is not a diagnosis but is a descriptive term when plasma glucose is above the “normal” range but does not meet the criteria for “hyperglycemia.” Persons with prediabetes are at increased risk for developing T2DM.
Diagnosing diabetes in symptomatic patients
Symptoms of DM need not be present to establish the diagnosis of DM. DM is diagnosed in asymptomatic individuals who are hyperglycemic according to at least 2 different parameters on a single day, or are hyperglycemic according to at least 2 parameters on 2 different days.
Hyperglycemic in 2 different parameters on a single day could include a FPG of =>126 mg/dL (=>7 mmol/L) and a 2h-PG of =>200 mg/dL (11.1 mmol/L) during an OGTT; a FPG of =>126 mg/dL (=>7 mmol/L) and a HbA1c of =>6.5% (=>48 mmol/mol); or a 2h-PG of =>200 mg/dL (=>11.1 mmol/L) obtained during an OGTT and a HbA1c of =>6.5% (=>48 mmol/mol). In persons being tested for DM on 2 different days, it is advised that the first abnormal test be the test that is performed on the second day. However, this is not an absolute requirement.
Notes on screening for diabetes mellitus
Screening for T1D is not recommended at the present time because prevention is not yet available. However, many studies are ongoing about how T1D can be predicted using islet autoantibodies [5] and prevented through immunomodulation [6].
Screening for T2D is described in detail in reference [1]. Adults who are overweight or obese, have a first-degree relative with DM, are a member of a high-risk racial or ethnic group, have a history of cardiovascular disease, are hypertensive, have elevated triglyceride levels or low HDL cholesterol levels, women with polycystic ovarian syndrome, persons with HIV infection, physically inactive adults and adults with any manifestation of insulin resistance should be screened. Screening can be performed by the measurement of a FPG, 2h-PG during an OGTT, or by measuring HbA1c. Persons with prediabetes should be screened yearly for T2D. Women with a history of GDM should be screened every 3 years for T2D. Otherwise, adults should be screened every 3 years for DM beginning at age 45 years. The guidelines for risk-based screening for prediabetes or T2D in children is described in reference 1, table 2.4.
Conclusion
It behooves laboratorians to stay up to date on the methods and cut points that are used to diagnose DM. While not discussed in this overview, the diagnosis of gestational diabetes mellitus certainly deserves periodic review and is included in reference [1]. As well, proper performance of the OGTT is important when the OGTT is used in diagnosing DM. Some scholars have suggested measuring the plasma glucose at 1 hour during the OGTT as a tool in identifying individuals at increased risk for DM [7]. At present, consensus is lacking on this proposal. Stay tuned ……
References
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2021. Diabetes Care. 2021 Jan;44(Suppl 1):S15-S33.
- Kitabchi AE, Umpierrez GE, Murphy MB, Barrett EJ, Kreisberg RA, Malone JI, Wall BM; American Diabetes Association. Hyperglycemic crises in diabetes. Diabetes Care. 2004 Jan;27 Suppl 1:S94-102.
- Nadeau KJ, Anderson BJ, Berg EG, Chiang JL, Chou H, Copeland KC, Hannon TS, Huang TT, Lynch JL, Powell J, Sellers E, Tamborlane WV, Zeitler P. Youth-Onset Type 2 Diabetes Consensus Report: Current Status, Challenges, and Priorities. Diabetes Care. 2016 Sep;39(9):1635-42.
- Edwards CM, Cusi K. Prediabetes: A Worldwide Epidemic. Endocrinol Metab Clin North Am. 2016 Dec;45(4):751-764.
- Winter WE, Schatz DA. Autoimmune markers in diabetes. Clin Chem. 2011 Feb;57(2):168-75.
- Herold KC, Bundy BN, Long SA, Bluestone JA, DiMeglio LA, Dufort MJ, Gitelman SE, Gottlieb PA, Krischer JP, Linsley PS, Marks JB, Moore W, Moran A, Rodriguez H, Russell WE, Schatz D, Skyler JS, Tsalikian E, Wherrett DK, Ziegler AG, Greenbaum CJ; Type 1 Diabetes TrialNet Study Group. An Anti-CD3 Antibody, Teplizumab, in Relatives at Risk for Type 1 Diabetes. N Engl J Med. 2019 Aug 15;381(7):603-613.
- Jagannathan R, Neves JS, Dorcely B, Chung ST, Tamura K, Rhee M, Bergman M. The Oral Glucose Tolerance Test: 100 Years Later. Diabetes Metab Syndr Obes. 2020 Oct 19;13:3787-3805.
Legend
Figure 1 – The approaches to the diagnosis of DM are outlined. See text for details.