Summary
DOI: 10.1373/clinchem.2014.235143
At his 6-month well baby visit, a male infant was noted to have splenomegaly without any other notable physical examination findings. His medical history was unremarkable. He was born at term by elective repeat Cesarean section, with normal growth and development.
Student Discussion
Student Discussion Document (pdf)
Sarah K. Anisowicz1* and Jude M. Abadie2
1 Department of Pediatrics and 2 Department of Pathology, Tripler Army Medical Center, Honolulu, HI.
* Address correspondence to this author at: Tripler Army Medical Center, 1 Jarrett White Road, MCHK-PE, Honolulu, HI 96859. Fax 808-433-4837; e-mail [email protected]. The views expressed in this case study are those of the authors and do not reflect the official policy of the Department of the Army, the Department of Defense, or the United States Government.
Case Description
At his 6-month well baby visit, a male infant was noted to have splenomegaly without any other notable physical examination findings. His medical history was unremarkable. He was born at term by elective repeat Cesarean section, with normal growth and development. A screening complete blood count and comprehensive metabolic panel were ordered, but laboratory testing could not be performed due to grossly lipemic samples (Fig. 1). At that point, the patient was referred to pediatric gastroenterology owing to splenomegaly with subsequent lipemic samples.
Physical examination by pediatric gastroenterology at 7 months described a large infant with weight in the 99th percentile (10.4 kg), 81st percentile for length (71 cm), and 70th percentile for head circumference (44.5 cm). Body mass index was in the 99th percentile. Growth charts indicated that he was born at the 60th percentile for weight and increased to the 99th percentile at 6 months. Since the time of birth, length and head circumference demonstrated a stable growth rate at 80th and 57th to 70th percentiles, respectively. Multiple lipomas on his left ear lobe, lateral canthus of the right eye, abdomen, and back were noted on examination. Abdominal examination showed a nondistended, soft, nontender abdomen with splenomegaly 2 cm below the lower costal margin. Hepatomegaly was absent, and no other abdominal masses were appreciated.
Family history was remarkable for a maternal grandmother who was diagnosed with hypertriglyceridemia and hypercholesterolemia discovered during an episode of pancreatitis. Triglycerides were reportedly “in the thousands” before treatment was initiated. Abdominal ultrasound was notable for splenomegaly of 3.4 by 8.3 cm (upper reference limit 6.5 cm). The
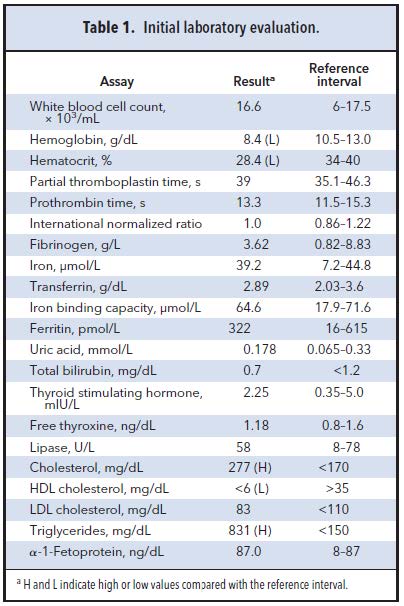
complete blood count listed in Table 1 suggested anemia and demonstrated a white blood cell count within reference intervals. Coagulation panel, fibrinogen, lipase, uric acid, and thyroid studies were all within reference intervals. The lipid panel demonstrated increased total cholesterol and triglycerides, with LDL within reference intervals and low HDL. VLDL concentrations could not be calculated because of the markedly increased triglycerides. α-1-Fetoprotein was within reference intervals for the patient’s age (1).
Lipoprotein phenotyping demonstrated increased triglycerides (661 mg/dL), low total cholesterol (85 mg/dL), low HDL cholesterol (5 mg/dL), and presence of chylomicrons with α and β band within reference intervals and increased pre-β band. VLDL and LDL were not calculated because of increased triglycerides. These results were indicative of type IV familial hyperlipidemia (isolated hypertriglyceridemia), and the patient was prescribed dietary fish oil supplementation. The patient’s parents underwent phenotyping screening as well, with the mother showing a normal lipid profile and the father a similar hypertriglyceridemia.
Reference
- Blohm ME, Vesterling-Hörner D, Calaminus G, Göbel U. Alpha 1-fetoprotein (AFP) reference values in infants up to 2 years of age. Pediatr Hematol Oncol 1998;15:135– 42.
Questions to Consider
- What laboratory markers help to navigate and guide a differential diagnosis in patients presenting with splenomegaly?
- How can lipemia affect laboratory test results?
Final Publication and Comments
The final published version with discussion and comments from the experts appears
in the December 2015 issue of Clinical Chemistry, approximately 3-4 weeks after the Student Discussion is posted.
Educational Centers
If you are associated with an educational center and would like to receive the cases and
questions 3-4 weeks in advance of publication, please email [email protected].
AACC is pleased to allow free reproduction and distribution of this Clinical Case
Study for personal or classroom discussion use. When photocopying, please make sure
the DOI and copyright notice appear on each copy.
DOI: 10.1373/clinchem.2014.235143
Copyright © 2015 American Association for Clinical Chemistry