Summary
DOI: 10.1373/clinchem.2010.155122
A 43-year-old man with epilepsy who had been treated with valproic acid (Depakote®) (VPA)2 for 13 years and had a history of multiple suicide attempts and alcohol dependence presented after ingesting an overdose of VPA and ibuprofen. The patient was not on any other prescribed medications. At presentation, the patient exhibited the following: Glasgow Coma Score (GCS), 3/15 (3 being the worst, indicating full coma, and 15 being a normal mental state); heart rate, 96 beats/min; blood pressure, 121/79 mmHg; temperature, 36.5 °C; oxygen saturation, 97%.
Student Discussion
Student Discussion Document (pdf)
Andrew S. Davison,* Anna M. Milan, and Norman B. Roberts
Department of Clinical Biochemistry and Metabolic Medicine, Royal Liverpool University Hospital, Liverpool, UK.
*Address correspondence to this author at: Department of Clinical Biochemistry and Metabolic Medicine, Royal Liverpool University Hospital, Duncan Bldg., Prescot St., Liverpool L7 8XP, UK. Fax +44-151-706-5813; e-mail [email protected].
Case Description
A 43-year-old man with epilepsy who had been treated with valproic acid (Depakote®) (VPA) for 13 years and had a history of multiple suicide attempts and alcohol dependence, presented after ingesting an overdose of VPA and ibuprofen. The patient was not on any other prescribed medications. At presentation, the patient exhibited the following: Glasgow Coma Score (GCS), 3/15 (3 being the worst, indicating full coma, and 15 being a normal mental state); heart rate, 96 beats/min; blood pressure, 121/79 mmHg; temperature, 36.5 °C; oxygen saturation, 97%.
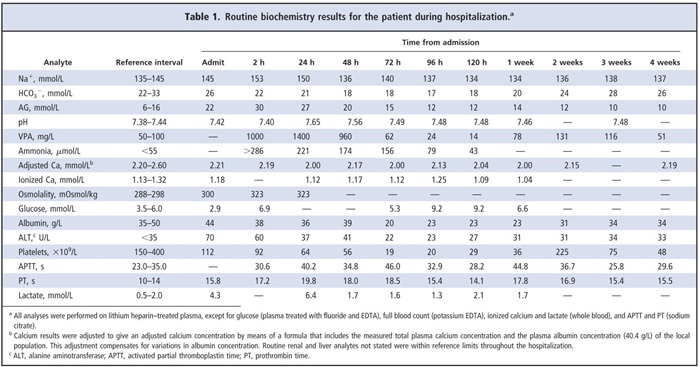
The patient’s plasma VPA concentration 3 h after admission was 1000 mg/L [therapeutic interval, 50–100 mg/L)] and increased to 1400 mg/L 14 h after admission. The amount of valproic acid ingested could not be determined from the case history, and it was not possible to accurately calculate the amount ingested because the absorption, the volume of distribution, and the elimination constant of VPA can be altered after an overdose. Paracetamol (acetaminophen), salicylate, and ethanol concentrations in blood were below their respective detection limits. The results of a computed tomography scan of the brain and an electroencephalography evaluation were normal. The amount of ibuprofen taken was unknown, and the plasma concentration was not measured.
Of particular note in the results of the admission biochemistry blood tests (Table 1) were an increased plasma osmolality (osmolal gap, <10 mmol/L on admission), an increased anion gap (AG), a low glucose concentration, and increased alanine aminotransferase activity. Blood gas results were unremarkable apart from an increased O2 partial pressure (PO2) 32.3 kPa (fraction of inspired oxygen, 50%) and an increased lactate concentration of 4.9 mmol/L. Additional blood investigations 2 h after admission showed severe hyperammonemia (ammonia, >286 μmol/L; upper reference limit, 55 μmol/L), hypernatremia, an increased plasma osmolality, and an increased AG (Table 1). The results of a urine screen for drugs of abuse were negative. The patient developed a profound respiratory alkalosis 6–10 h after admission secondary to the hyperammonemia.
Initial treatment involved standard intravenous rehydration with Hartmann solution (131 mmol/L Na+, 5 mmol/LK+, 2 mmol/L Ca2+, 29 mmol/L lactate, and 111 mmol/L Cl–), naloxone because of his reduced consciousness, and N-acetylcysteine as a precautionary measure. Thepatient was then transferred to the intensive care unit, where he received a 6-g infusion of L-carnitine over 16 h (approximately 100 mg/kg), with the aim of normalizing his ammonia concentration (as recommended by the UK National Poisons Unit). Hemodialysis was performed to aid in VPA removal. The initial attempt was unsuccessful because the patient had poor venous access. At 36 h after admission, hemodialysis removed 0.5 L of fluid, and an additional hemodialysis session (63 h after admission) removed another 0.5 L of fluid. The patient’s recovery was slow, with aGCSof 6/15 by day 4 and further improvement to 12/15 by day 6. He received 7 days of total parenteral nutrition. During this initial period, he developed thrombocytopenia and mild hypocalcemia (Table 1). The hypocalcemia was treated with Calcichew D3 Forte (2 tablets daily; Shire Pharmaceuticals).
A number of pseudoseizures were documented over several weeks. VPA therapy was briefly reintroduced 7 days after admission when plasma concentrations were below the therapeutic interval, but it was discontinued 17 days later because of the recurrence of thrombocytopenia. Lamotrigine was then introduced at a starting dosage of 25 mg once daily. The dosage was then titrated over a 2-week period to 100 mg daily (as an outpatient). The patient was discharged after 32 days.
Questions to Consider
- What was the cause of the increased AG observed in this patient?
- Why do individuals taking VPA develop increased ammonia concentrations?
- What was the cause of hypernatremia in this patient?
- Is it beneficial to measure free VPA or VPA metabolite concentrations in overdose cases?
Final Publication and Comments
The final published version with discussion and comments from the experts appears
in the September 2011 issue of Clinical Chemistry, approximately 3-4 weeks after the Student Discussion is posted.
Educational Centers
If you are associated with an educational center and would like to receive the cases and
questions 3-4 weeks in advance of publication, please email [email protected].
AACC is pleased to allow free reproduction and distribution of this Clinical Case
Study for personal or classroom discussion use. When photocopying, please make sure
the DOI and copyright notice appear on each copy.
DOI: 10.1373/clinchem.2010.155122
Copyright © 2011 American Association for Clinical Chemistry