Though encephalitis still is rare, its incidence both in the United States and internationally is increasing along with a rise in viral diseases like herpes, West Nile disease, Japanese encephalitis virus, and tick-borne encephalitis virus, among other causes. Even in the face of this growing menace, however, lack of consensus on case definitions, standardized diagnostic approaches, and priorities for research have blocked progress in combatting the disease. As a consequence, the process of diagnosing encephalitis is highly variable among clinicians and institutions, and fraught with inappropriate testing.
In response, the International Encephalitis Consortium (IEC), a multidisciplinary committee begun in 2010, recently issued a comprehensive guideline, which proposes standard definitions and a diagnostic algorithm for evaluating both children and adults with suspected encephalitis worldwide (Clin Infect Dis 2013;57:1114–28). “We anticipate that this algorithm can lead to rational, as opposed to shotgun, testing in cases of suspected encephalitis,” said the guideline’s first author, Arun Venkatesan, MD, PhD, assistant professor of neurology and director of the Encephalitis Center at Johns Hopkins University in Baltimore.
Noting that clinicians’ desperation to help critically ill patients drives inappropriate use of many tests, the guideline’s senior author, Carol Glaser, MD, said the document can augment the role of laboratorians, who are often “the voice of reason” tasked with advising physicians about suitable assays for encephalitis patients. Glaser is chief of the Encephalitis and Special Investigations Section of the California Department of Public Health (CDPH) and former principal investigator of the California Encephalitis Project, a partnership between CDPH and the U.S. Centers for Disease Control and Prevention (CDC).
Encephalitis, the guideline notes, remains steeped in mystery, its causes ill-defined and progress unpredictable. A plethora of viruses, bacteria, and autoimmune diseases can lead to brain inflammation, but more than half the time, the precise culprit remains unknown. Symptoms of encephalitis range from fever, headache, and confusion to seizures, severe weakness, and even language disability. The condition takes a disproportionate toll on the very young and elderly, who often are among the sickest patients, languishing for months in intensive care units. Clinicians become desperate to keep these very ill patients from joining the ranks of severely disabled encephalitis survivors, or worse, the disturbingly large proportion who die from the condition.
A Comprehensive Reference
The guideline is notable not only for its wide scope and intended ease of use but also for the new recommendations it makes, said Glaser. Other encephalitis guidelines have made similar recommendations, but targeted specific types of infections or age groups, she added. Glaser, who worked on previous encephalitis guidelines issued by the Infectious Diseases Society of America, noted that they were also quite comprehensive, but “were too unwieldy to use at the bedside.” In contrast, the IEC guidelines include an algorithm the authors hope will make them more useful to clinicians.
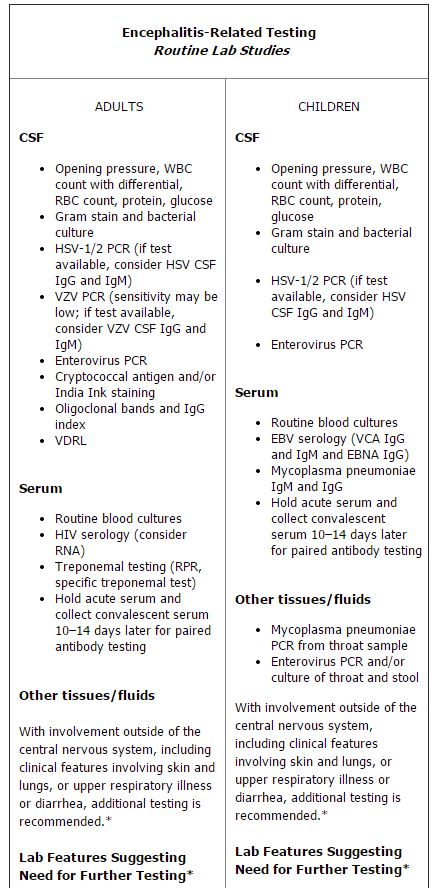
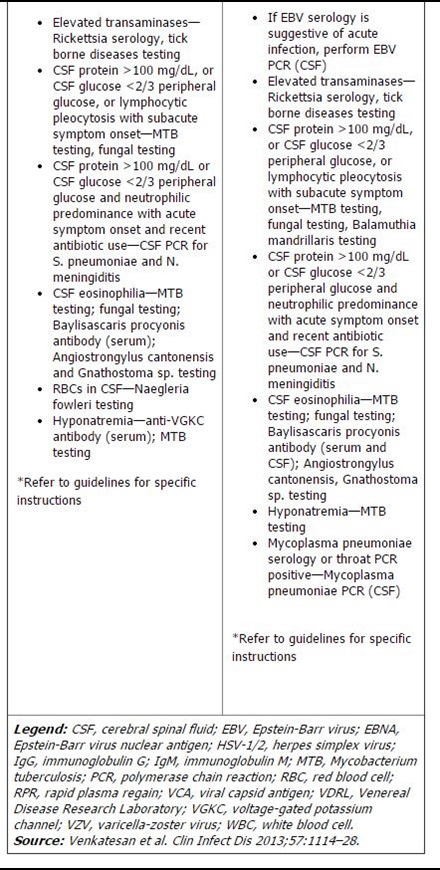
The algorithm in question lists routine studies and tests that should be performed on all suspected encephalitis patients, as well as conditional studies based on host and geographic factors, season and exposure, specific signs and symptoms, and both laboratory and neuroimaging features (See chart, p. 4). The guideline also specifies which tests are appropriate for children, and which should be used in adults only.
This stratified approach of performing a clutch of routine tests plus conditional studies—based on patient-specific circumstances—is a pragmatic approach to the diagnostic work-up of encephalitis, given that many of the recommended tests use precious cerebral spinal fluid (CSF), said James Sejvar, MD, a neuroepidemiologist in the Division of High Consequence Pathogens and Pathology at CDC’s National Center for Emerging and Zoonotic Infectious Diseases in Atlanta. “You can’t get much CSF, so you have to be selective about how you use it,” he explained.
Another strength of the IEC document is its inclusiveness. In contrast to previous recommendations for diagnosing encephalitis which came from individuals or institutions, this guideline was written as a consensus by 23 authors representing multiple medical specialties, countries, and institutions, Sejvar pointed out.
The guideline also sets out specific criteria for distinguishing encephalitis from encephalopathy, terms often used interchangeably in the medical literature because of their overlapping sets of symptoms. But the IEC document clearly defines the two conditions, noting that encephalopathy refers to any dysfunction of the brain, which can be caused by many different conditions, including encephalitis. In contrast, encephalitis refers specifically to brain inflammation as confirmed via histopathology or on the basis of selected clinical, laboratory, electroencephalographic, and neuroimaging features. The distinction is important, noted Sejvar. “In various scenarios, people define encephalitis differently, and this makes it difficult to compare studies and draw conclusions about the disease,” he explained.
The IEC guideline newly suggests three studies as routine, said Glaser. These include enterovirus polymerase chain reaction (PCR), oligoclonal bands, and immunoglobulin G (IgG) index in CSF. Enteroviruses are RNA viruses that typically replicate in the gastrointestinal tract. The viruses can spread to other organs, including the brain, causing severe encephalitis. A positive result on PCR indicates the presence of viral nucleic acid. The presence of oligoclonal bands in CSF, combined with their absence in blood serum, can also mean that immunoglobulins are produced in the central nervous system. Neurologists on the IEC panel suggested these tests, Glaser noted.
A Role for Laboratorians
Laboratorians can help clinicians implement the guidelines by advising them on how to better use and interpret results of recommended assays. This is particularly true with PCR assays, which often produce false-positive or false-negative results, guideline authors told CLN. False-negatives can occur in the presence of inhibitory substances that interfere with the PCR reaction, low viral load, or insufficient amounts of the target in patient samples, Glaser explained. False-positives can enter the picture from contamination in the laboratory, coincidental detection of the agent in question when it isn’t the cause of the current infection, or from pushing a PCR protocol beyond its limits of detection. The latter can result in amplification of spurious products such as fragments of human DNA instead of the intended target, Glaser noted.
Clinicians, Glaser and others said, often rely too heavily on PCR to diagnose causes of encephalitis, without realizing its limitations. “Molecular tools are wonderful, but you have to know how to use them,” Glaser said.
Despite the challenges presented by PCR, the guideline recommends PCR tests for herpes simplex virus (HSV) and varicella-zoster virus (VZV), all the while cautioning about its pitfalls and the need for additional testing. For example, PCR for HSV can be negative early in the course of the disease. Because of this, the IEC panel recommended repeating this test with a new CSF sample from a second puncture within 3–7 days if the initial results are negative and the clinician still suspects herpes encephalitis. Many clinicians rely too much on a single CSF sample, according to Glaser. “If the diagnosis still is unknown a few days after the first CSF was obtained, a second sample might be quite helpful, particularly for re-checking herpes PCR.”
Laboratorians can also remind clinicians that PCR testing should be augmented with other assays. This is the case when testing for VZV, a leading cause of encephalitis. The guideline calls for both PCR and antibody testing for VZV because detection of antibodies to VZV in CSF appears to have greater sensitivity than detection of viral DNA, Venkatesan explained. “The same can be said of arboviruses such as West Nile virus, where the virus is often not detectable by PCR in the serum or spinal fluid once symptoms of encephalitis have developed,” he added. “Thus, antibody testing is crucial in such situations.”
Serology: No Silver Bullet
As important as serology-based testing for antibodies is in the work-up of suspected encephalitis, it also presents challenges to labs and requires caution, noted both Venkatesan and Sejvar. “The message to labs is that for certain agents, initial serological results may require more specific follow-up tests,” said Sejvar. This is particularly true for arboviruses such as West Nile virus, which can cross-react with antigens from other arboviruses. Additional analysis such as plaque reduction neutralization testing can improve specificity, according to Venkatesan. In the case of mycoplasma, because it is a ubiquitous agent associated with a substantial baseline infection rate, a positive immunoglobulin M (IgM) test may simply indicate concurrent respiratory infection rather than implicating mycoplasma as a cause of encephalitis. “West Nile virus IgM can persist for many months after infection, and therefore a positive result does not necessarily indicate active West Nile infection,” explained Venkatesan.
The pitfalls of serology are aptly illustrated in tests for Lyme disease, according to Sejvar. He noted that although the guideline does not specifically address the disorder, encephalitis caused by the culprit bacterium, Borrelia burgdorferi, is a particular concern in some areas of the U.S. CDC recommends that if an enzyme-linked immunosorbent assay (ELISA) yields a positive result, it should be confirmed with serology by Western blot analysis to avoid inappropriate, long-term treatment, Sejvar emphasized.
Another issue with Lyme disease testing “is interpreting a positive test as meaning active disease,” said Stephen Gluckman, MD, medical director of Penn Global Medicine and professor of medicine at the Perlman School of Medicine at the University of Pennsylvania in Philadelphia. “These antibodies could have been present for years.”
The guideline also recommends corresponding CSF and serum testing for human herpes virus 6 in patients with prominent memory, learning, and emotional symptoms. Research by IEC panelist Katherine Ward has shown that roughly 1% of people worldwide carry the virus in their genomes, but that its levels in CSF due to primary infection differ from those present because of integration of the virus into chromosomes (J Clin Microbiol 2007;45:1298–304). However, latent disease may also confound results, according to the IEC document.
The authors also call for testing anti-N-methyl-D-aspartate receptor (NMDAR) in CSF if a serum test is negative and there is still clinical suspicion for this entity, Venkatesan noted. While anti-NMDAR receptor encephalitis is typically diagnosed by clinical symptoms and a positive antibody in the serum, “recent literature suggests that in up to 15 percent of individuals, the antibody may only be present in the CSF and not in the serum,” he explained.
Rabies testing, which the guidelines suggest for patients who have had animal bites, is often riddled with confusion, Glaser observed. “This test should be done only at a public health lab because it entails a lot of nuance,” she said, explaining that rabies tests done at commercial labs don’t diagnose human rabies. Rather, they determine whether a patient who had a rabies vaccine is immune to the virus, a point lost too often in clinical practice, according to Glaser. “I see samples inappropriately sent to commercial labs for rabies testing all the time.”
Gluckman, who did not serve on the IEC panel, cautioned about placing too much stock in any one test result for encephalitis. “There’s no perfect test,” he said. “There are false-positives and false-negatives for every test. You still need to look at patients and decide if the data make sense. Diagnosis requires thinking.”
Too Much? Not Enough?
While the IEC guideline is intended to reduce unnecessary testing, Gluckman disagreed with the breadth and scope of the more than 25 tests and studies recommended as routine by the algorithm. Noting that he usually first tests suspected encephalitis patients for herpes, varicella, and HIV, and depending on the season, arboviruses such as West Nile and enteroviruses, Gluckman said that rather than automatically ordering all the studies the IEC guideline lists as routine, he would look to the document when first-line tests fail to pinpoint a cause. “Clinical skills and judgment should temper testing,” he suggested. Still Gluckman said he found the guideline to be a very useful, comprehensive compendium of information about identifying known causes of encephalitis. “This is a great resource listing all the tests you might consider,” he added.
It might be acceptable to skip some tests if clinical evidence points strongly to a specific cause, Glaser agreed. “If a clinician is pretty sure it’s herpes, for example, he could go for that and hold other tests in reserve. But if there’s no big clue, really he should go for all of the routine tests,” she explained, noting that sometimes, one test may indicate the need for another. For example, she said, imaging results sometimes drive herpes testing.
The number of recommended routine tests isn’t particularly large compared to what Glaser, an infectious disease specialist who previously served as the medical officer for CDPH labs, has seen many physicians order. “It’s puny compared to what some patients get,” added Glaser, recalling that she commonly saw clinicians order as many as 50 tests for an individual patient. “So this list isn’t over the top at all.”
Glaser was careful not to fault clinicians for inappropriate testing decisions. If they order a battery of tests that turn out to be unnecessary, it’s only because they’re in a race to diagnose and treat sometimes desperately ill patients, she suggested.
Looking to the Future
As the epidemiology of encephalitis changes, research yields new causes, and testing modalities change, so will the algorithm, Venkatesan said. “We view the published algorithm as a starting point,” he explained, adding that the IEC is now developing a living version of this algorithm that it plans to update and make available online as new information regarding encephalitis emerges.
In light of the changing clinical and scientific picture of encephalitis, the authors also proposed research priorities, including a better understanding of genetic risk factors for the disease which one day might explain pathogenic mechanisms, define relevant biomarkers, and suggest potential therapeutic approaches. Researchers also need a more complete view of several leading causes of encephalitis such as arboviruses, Japanese encephalitis virus, tick-borne encephalitis virus, and tick-transmitted Powassan virus, among others.
While this research might identify new roles for laboratorians in the future, for now, they can make an important contribution to the field by familiarizing themselves with the IEC document, said Glaser. “If you see a problem in how tests are ordered, reach out to physicians. Help them by suggesting more appropriate tests,” she suggested.
In those institutions that regularly include laboratorians in patient rounds, that advice can be offered at the bedside, along with information about quality of tests, the best samples to use, and how to store them, said Gluckman, who sees hospitalized patients along with both a laboratorian and pharmacist. “It’s amazing how helpful laboratorians are,” he said. “They can talk about the quality of tests, and give useful information about how to best obtain and store samples.”
Deborah Levenson is a freelance writer who lives in College Park, Md. Her email address is [email protected].