
It takes hard work for labs to maintain good communication with clinicians and help them understand results. Now, the Department of Health and Human Services (HHS) thinks it's time for labs to get cozy with patients, too. Under a proposed rule currently being finalized, all labs across the country will be required to send results directly to patients upon request, ending a patchwork system of state laws and federal regulation that prevents this in more than half of states. In order to clear a direct line from labs to patients, the proposal makes changes to both the Clinical Laboratory Improvement Amendments (CLIA) and the Health Insurance Portability and Accountability Act (HIPAA).
As a handful of government agencies work out the details of the final regulation, the implications of this change have begun to sink in, with laboratorians and many in the healthcare community concerned about unintended consequences. A few large labs and other healthcare providers with cutting-edge technology already offer patients rapid, online access to their lab results in certain states. But the HHS plan for direct patient access could catch many labs off guard, according to Joshua Freemire, an attorney with Ober-Kaler.
"There are going to be a lot more compliance issues than people realize, and it's going to take a fair amount of work to get all the policies, procedures, and workflow in place," Freemire said. "Labs should really be paying close attention to this because once the rule is final; there will not be much time to comply." Freemire advises physicians, hospitals, pharmacies, laboratories, and other healthcare providers on regulatory compliance, licensing, and reimbursement for the firm's health law group.
From Zero to 60
Once HHS publishes final regulations, timing will be critical. The proposed rule gives labs 180 days after the effective date of the final rule to comply. Even without a final rule in hand, labs should begin some level of preparation immediately, according to Freemire. "My biggest concern if I were in a lab would be to get started on policies and procedures to address this now so that I have something in place," Freemire said. "That way, I could do any necessary training or workflow adjustments within the prescribed timeframe following the finalization of this rule." Currently, only nine states have laws explicitly allowing test results to go directly to patients (See Box, below).
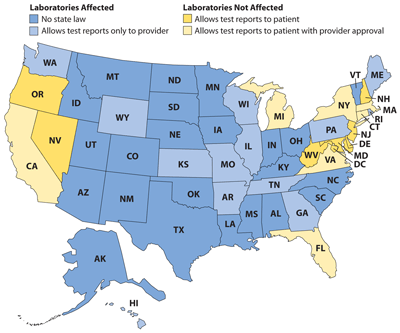
Sending Results Directly to Patients Will Override State Laws
The proposed rule granting patients direct access to their lab results will overrule laws in 13 states that prohibit such access. Including the 26 states and territories that have no law defining who can receive direct access to results, a total of 39 states and territories will be affected. Nine states allow results to go directly to patients, and seven allow patient access after provider approval.
In the proposed rule, HHS estimates that it will take labs 2–9 hours to develop their policies and procedures for direct patient access. In comments submitted to HHS, Alan Mertz, president of the American Clinical Laboratory Association (ACLA), strongly disagreed with this assessment. "There are several reasons why compliance in such a short period of time will be difficult at best," he wrote. "Some laboratories lack policies, protocols, and mechanisms for responding to requests for access to test reports, for addressing follow-up questions from patients, or for providing patients with direct access to test reports. Such laboratories will have to establish new procedures and new software systems to accommodate the proposed changes. They also will have to develop policies and procedures for verifying an individual's identity." Staff training, as well as concurrent regulatory changes such as meaningful use of electronic health records (EHR), will also make a quick patient access transition difficult.
A significant hurdle for labs could be verifying the identity of the patient requesting his or her information, Mertz emphasized. Labs rarely have direct contact with patients, and so must rely on providers for patient information. "The patient's contact information and demographic information often are provided to the laboratory by the healthcare provider ordering the test, and sometimes that information is inaccurate or incomplete," Mertz wrote to HHS. "Furthermore, laboratories usually do not have access to a patient's medical chart and usually are unaware of the patient's diagnosis, comorbidities, and medical history." While expressing overall support for the concept of patient access to lab results, Mertz urged HHS to give labs a full year to come into compliance once the rule is finalized.
One of the reasons that Mertz and others are expressing concern about correctly identifying patients is that compliance with this rule means sharing more than just individual test reports with patients—labs must be ready to hand over all the information the lab has on that person. As a result, the proposal will bring changes even for the nine states where state law already allows test reports to go directly to patients (See Box, right). "Even if labs started preemptively to mail a copy of test results to everyone that would not comply with HIPPA requirements under this proposed rule," Freemire said. "The lab must be ready to include all of the health information they maintain about the patient. If it's a new patient, that may be very limited, but if it's someone that's been to the lab many times before, it may be a more sizable file."
|
Direct Patient Access Proposal Requires Changes to CLIA, HIPAA
Under the Department of Health and Human Services (HHS) proposal to allow direct patient access to lab results, two areas of law familiar to labs will require significant alterations: the Clinical Laboratory Improvement Amendments (CLIA) and the Health Insurance Portability and Accountability Act (HIPAA).
Under the current regulations, CLIA only allows labs to disclose test results to an authorized person or referring lab. CLIA defers to state law to define who an authorized person is. HIPAA gives patients the right to inspect and obtain a copy of their protected health information, but the Privacy Rule included in HIPAA exempts CLIA-covered entities. Patching both laws will allow direct patient access to lab results in all states, regardless of state law.
However, according to attorney Joshua Freemire, labs must keep in mind that if they will no longer be exempt from the HIPPA provisions excluding lab data, it may not only be lab results that patients have a right to access. It could include any and all data the lab maintains on the patient. This is one reason that labs are eagerly awaiting the details for this area in the final rule, even in those states that currently allow patients to be an authorized person under CLIA.
"This proposed rule is different in that what those states permit is releasing of test results to patients," said Freemire. "Once the exception no longer exists in the HIPPA Privacy Rule, there needs to be a means by which patients can make a formal request for their protected health information, and the information labs will need to provide may not be limited to test results." Freemire advises physicians, hospitals, pharmacies, laboratories, and other health care providers on regulatory compliance, licensing, and reimbursement for the Ober-Kaler health law group.
The change to HIPAA is made even more complicated by the fact that the Privacy Rule is currently in flux. HHS proposed a revised Privacy Rule that has yet to be finalized. According to Freemire, the updated Privacy Rule could change the format in which labs will have to provide health information. For example, it may be required to be available electronically and within a shorter timeframe.
|
Are Lab-Savvy Patients Healthier?
HHS Secretary Kathleen Sebelius has made it clear that giving patients direct access to their lab results is aimed at improving care. "When it comes to healthcare, information is power," Sebelius said when she first announced the proposed rule last September. "When patients have their lab results, they are more likely to ask the right questions, make better decisions, and receive better care."
Exactly how patients independently armed with lab data will go on to achieve better outcomes is uncertain, however. In certain circumstances, such as self-monitoring with blood glucose meters, research does demonstrate benefits for patients. However, opening up lab reports may prove to be quite different. In a commentary published in the Journal of the American Medical Association in December, Hardeep Singh, MD, MPH, and his colleague Traber Davis Giardina, MA, MSW, pointed out that up to 26% of abnormal test results are not followed-up in a timely manner, making the HHS proposal seem like a step in the right direction on an important patient safety issue. At the same time, Giardina and Singh, both researchers at the Houston Veteran Affairs Health Services Research and Development (HSR&D) Center of Excellence, found little consensus in the literature and conflicting empirical evidence to support a conclusion on direct patient access to abnormal test results.
When it comes to lab results, the empirical link between empowered patients and better health outcomes is far from clear. In fact, Singh's research on how physicians share responsibility for test results suggests an unexpected possibility: follow-up could deteriorate rather than improve. Singh published research in 2009 reporting that follow-up was worse when two clinicians—such as the primary care provider and the test ordering clinician—were notified of an abnormal test result. For example, a specialist might order a test but not follow-up on the abnormality because he or she expected the primary care provider to do so. But at the same time, the primary care provider expected the specialist to follow-up since the specialist ordered the test. The result: no one followed-up.
Much of this comes down to responsibility, according to Singh. When responsibility becomes distributed among several clinicians, or possibly between a clinician and a patient, confusion becomes more likely over whose job it is to follow-up. "There are a lot of these soft concepts that we often don't account for in healthcare," he said. "With issues such as responsibility diffusion and role confusion, clearly we're still learning when it comes to teamwork in healthcare. Overall, the movement is toward more transparency, empowerment, and engagement for patients, which I think is a good thing. But how we get there, and what kind of barriers we have to overcome, remains to be seen." Singh is an assistant professor of medicine at the Michael E. DeBakey Veterans Affairs Medical Center and Baylor College of Medicine, chief of the Health Policy and Quality Program at the Houston VA HSR&D Center of Excellence, and director of the Houston VA Patient Safety Center of Inquiry.
It is significant that the proposed rule keeps silent on which test results might be excluded or delayed due to potential anxiety and confusion for patients. States that currently allow direct access to lab results have provisions that require a waiting period or provider notification in order to make sure that sensitive results, such as a cancer diagnosis, come from the patient's physician. Singh also pointed out another unknown: how will patients respond to abnormalities that are not clinically significant? He noted that existing reimbursement models usually do not cover extra phone calls or face-to-face appointments with patients in order to explain lab results.
Lab consultant Paul Epner also underscored the need to improve follow-up, but he worried that if physicians often struggle to understand lab tests, patients might not do much better. "From a patient perspective, there are too many who are told that ‘no news is good news,' which isn't always true," Epner said. "Some literature shows that physicians fail to follow-up on test results." In addition, Epner mentioned what he calls "orphan test results"—results issued after hospitals discharge patients that no one reviews.
At the same time, laboratorians are all too familiar with physicians's often limited understanding of the lab, Epner said. "I don't think physicians have a very solid understanding of all the variables that can impact results," he said. "If the clinician doesn't appreciate all those factors, I worry that patients will have even less of a chance of understanding them."
As physicians and laboratorians debate the effect of giving patients greater ownership of their lab results, HHS clearly has patient empowerment and patient engagement in mind for the future. In the proposed second stage of HHS's meaningful use incentive program for EHRs announced in February, providers will be required to offer patients the ability to view online, download, and transmit their health information. Providers could be required to meet these goals by as early as 2015 or risk cuts in Medicare payments.
Seeking Out Patients
With the federal government's focus on driving adoption of EHRs, certainly the future of patient access to the lab will be electronic. Currently, for those states that don't allow direct access to lab results, patients must get this information from their physician. But most physicians are way behind. A 2011 Deloitte Center for Health Solutions survey of physicians, Physician Perspectives about Health Information Technology, found that only about half of all physicians use the Internet to inform or engage patients, and only 20% provide online scheduling or downloading of test results.
Meanwhile, at least one lab and several health systems have pushed ahead to offer rapid, if not direct, patient access to lab results. Quest Diagnostics now has some 130,000 users of its Gazelle software that allows patients to browse their lab results on their smartphones. Launched in 2010, the system gives physicians a 48 hour window to contact patients before results become available through the mobile app, and it blocks cancer and genetic test results. Quest offers the service in 33 states where either patient access is specifically allowed, or where the law is silent. However, even in states that prohibit direct access, physicians using Quest's Care360 cloud-based EHR can first review results and rapidly release them to patients. Quest developed its own in-house library of patient-oriented information about lab tests so that the app can offer brief explanations of results.
The Gazelle app is part of Quest's mobile-first strategy for patient engagement, according to Neil Desai, Quest's executive director of enterprise architecture. The company does not even offer a companion website to access results from personal computers. "We really believe that if you want patient engagement, you need to have a mobile solution," said Desai. "Our focus is on educating patients, getting them involved, and helping them manage an extremely complex healthcare system, and mobile delivers that." In addition to delivering lab test results wirelessly, the app includes other functions such as medication reminders and storage of other personal health information. These features require users to key in their information.
In a similar move, Kaiser Permanente went mobile in 2011, offering lab results, as well as the rest of their full-featured EHR, via smartphone to Kaiser's nearly 9 million patients. Like Quest, lab results arrive after a 48 hour delay. "Connectivity is the hallmark of the healthcare delivery system of the future," said George Halvorson, chairman and chief executive officer of Kaiser Permanente, in a statement before the Healthcare Information and Management Systems Society annual conference in February. "Kaiser Permanente is leading the way by making health and wellness tools available from anywhere in the world. Now, Kaiser Permanente patients can simply pull out their mobile devices and have immediate access to their own medical information, proving that Kaiser Permanente is the model for connected care. We have had one million page views of the content through smart phones in just the first month of availability. Members love being connected with their own health data and their own caregivers."
Research from the Pew Research Center's Internet and American Life Project released in March shows consumers adopting smartphones at a very rapid pace. Nearly half of American adults were smartphone owners as of February 2012, an increase of 11 percentage points over last year. Moreover, 71% of adults between 25–34 years own a smartphone.
Yet, access to lab results on traditional personal computers is nothing new, at least for about 200,000 Geisinger Health System patients in Pennsylvania. The health system has offered online access via its MyGeisinger EHR web portal for close to a decade. Even though state law does not allow direct patient access to test results, patients can still see this data because of rapid provider review and release. In other words, even though it's technically not direct, it feels that way to patients.
When Geisinger first launched the online EHR, it surveyed patients and found lab results at the top of their wish lists as an output from such a system, according to Jay Jones, PhD, director of regional laboratories at Geisinger. "Lab results go through the system very quickly," Jones said. "Once we enter the result in the lab information system, it almost immediately goes to the inbox of the clinician. If the result is normal, a physician okays it and it's available to MyGeisinger, usually in 24 hours or less. In many cases, if a patient has a morning appointment, by the time the person gets home he or she can look up the lab result." If patients have questions, they don't call the lab. MyGeisinger offers a messaging service for patients to query a nurse or a physician.
Even though patients strongly desire access to lab results, they still value medical supervision, according to Jones. "Having a physician review their results is very important to patients," Jones said. "And if results are abnormal or they have questions, they much prefer to talk to their doctors who know their medical situations."
Labs Need to Get it Right
According to Epner, HHS's proposal presents both opportunities and risks to labs. "On the opportunity side, we have an opening here to improve the awareness of the work that we do in the lab," he said. "However, I think we have a lot of work to do before we're ready to handle patient requests. And even if we direct patients to call physicians with questions, you must assume that some people will call the lab, and so you need to be prepared for that." Epner, who developed the Labs Are Vital public awareness campaign during his tenure as director of health care improvement initiatives for Abbott, worries that if labs have problems with sending results to patients, it could backfire and squander the potential to boost respect for the lab. "It could improve the relationship between patient and lab, which is virtually nonexistent now," he said. "But only if we're able devote the time and resources to do this right."